|
11/19/2023 0 Comments Anomeric carbon in straight chain 
This term is mostly used with organic compounds. Chirality refers to the property of having a superposable mirror image. 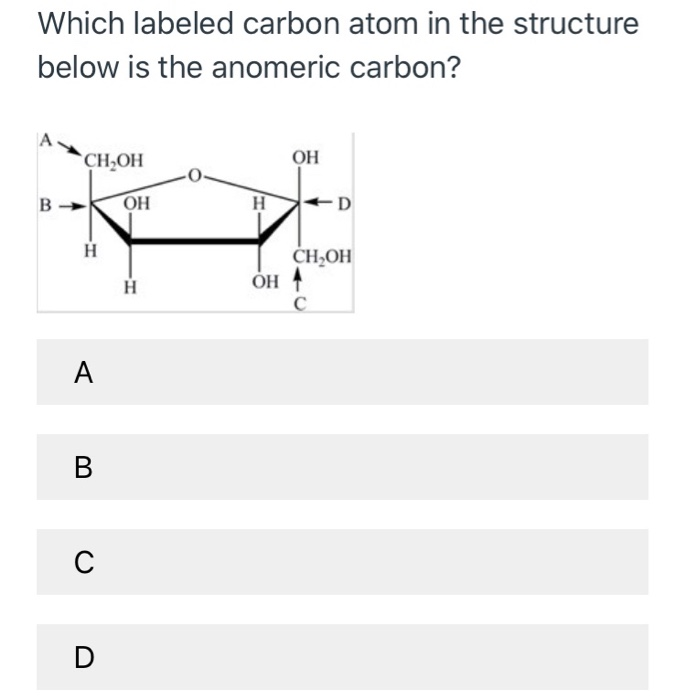
The concept of the presence of a chiral carbon is best described by chirality. What is Chiral Carbon?Ĭhiral carbon atoms are carbon atoms consisting of four single bonds to four different functional groups. That means the hydroxyl group and the exocyclic oxygen atom are on the opposite sides of the molecular projection. The following example shows the alpha anomer of D-glucopyranose.īeta anomer is the configuration of a carbohydrate in which the hydroxyl group is trans to the exocyclic oxygen at the anomeric centre. When we draw a Haworth formula, the hydroxyl group is in the downward direction if it is the alpha anomer. That means the hydroxyl group and the exocyclic oxygen atom are on the same side of the molecular projection. According to the chemical structure of anomeric carbon-containing compounds, there are two types as alpha anomers and beta anomers.Īlpha anomer is the configuration of a carbohydrate in which the hydroxyl group is cis to the exocyclic oxygen at the anomeric centre. Summary – Anomeric Carbon vs Chiral Carbon What is Anomeric Carbon?Īnomeric carbon is the carbon derived from the carbonyl carbon compound of the open-chain form of the carbohydrate molecule. Anomeric Carbon vs Chiral Carbon in Tabular Formĥ. Chiral carbon atoms are carbon atoms consisting of four single bonds to four different functional groups. ↑ Solano Community College (2007) Drawing Haworth Projections.The key difference between anomeric carbon and chiral carbon is that an anomeric carbon atom essentially contains a hydroxyl group that is either cis or trans to the exocyclic oxygen atom, whereas a chiral carbon atom essentially contains four different functional groups attached to it with single covalent bonds.Īnomeric carbon is the carbon originated from the carbonyl carbon compound of the open-chain form of the carbohydrate molecule.↑ Russel KC (1999) Haworth Projections.↑ Professional Education,Testing and Certification Organization International (2012) Organic Chemistry 16.4 Cyclic Structures of Monosaccharides. 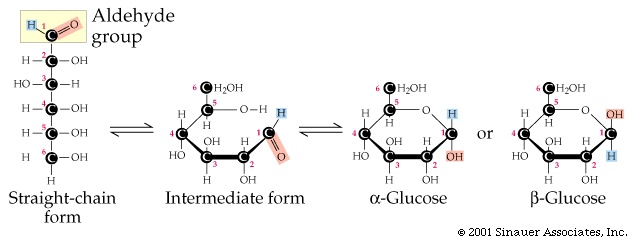
↑ Cornell College (2012) Reactions of monosaccharides.(2012) Biochemistry, 7th Edition, New York: Kate Ahr Parker. Ī Condensation reaction catalyses the formation of disaccharides between 2 monosaccharides with the elimination of a water molecule.ĭetermining which anomer is alpha and which is beta is easy when looking at the Haworth projection alpha anomers have different stereochemistry at carbon 1 and carbon 5, whereas beta anomers have the same stereochemistry at carbon 1 and 5. Under a dynamic equilibrium, 6-carbon pyranose rings are usually formed as compared to 5-carbon furanose rings. Anomers in the form of Fischer projection (open-chain sugar) makes up less than 1% of the total solution. The percentages of each type of anomers present are as follow: 25% are alpha anomers and 75% are beta anomers. The reaction is said to be at a dynamic equilibrium, where the rate of forward reaction is equal to the rate of backward reaction. In addition, these straight chain sugars are converted back into its ring structure. Through contact with water, the sugar monomers appear to be opening up their ring structure to form straight chain sugars. In the process of mutarotation, a solution of both anomers are heated at a high temperature, giving rise to individual monomers. Īlpha and beta anomers are formed from an open-chain sugar by mutarotation. It is possible to distinguish the two by the directions that the molecule rotates under plane-polarise light. A beta-glucose has its -OH parallel to the ring. An alpha-glucose has its -OH perpendicular to the ring. Thus, the carbon is called an anomeric carbon. They are identified by the direction that the -OH group is pointing to on the first carbon (C1) on the cyclic sugar. 
There are 2 forms of anomers, namely alpha and beta. As the name suggests, they are isomers of each other. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
